An electric cell converts chemical energy into electrical energy to produce electricity. It contains two electrodes immersed in an electrolyte called Anode and Cathode. A number of electric cells are connected together to form a battery.
When a cell or battery is connected to a circuit, electrons flow from the negative terminal to the positive terminal through the circuit. By using chemical reactions, a battery produces a potential difference across its terminals and this cell is called the Electrochemical cell.
In an Electrochemical cell, the chemical energy is converted into electric energy and vice versa. This potential difference provides the energy to move the electrons through the circuit. The starting point to the electric cell is the experiment by Luigi Galvani and his wife Lucia on the dissected frog hung from the iron railing with brass hooks.
Primary cells are one in which the electric energy is derived by irreversible chemical action is called Primary cells.
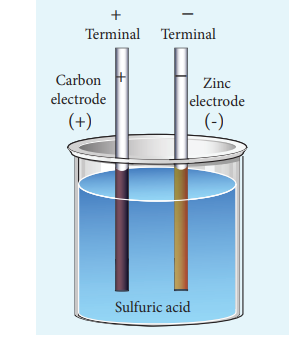
Voltaic Cell
- The simple cell or voltaic cell consists of two electrodes, one of copper and the other one is zinc dipped in dilute sulphuric acid in a glass vessel.
- The anode is copper.
- Cathode is zinc
- The potential difference is 1.08 V
- The electrolyte is dilute Sulfuric acid.
Daniel Cell
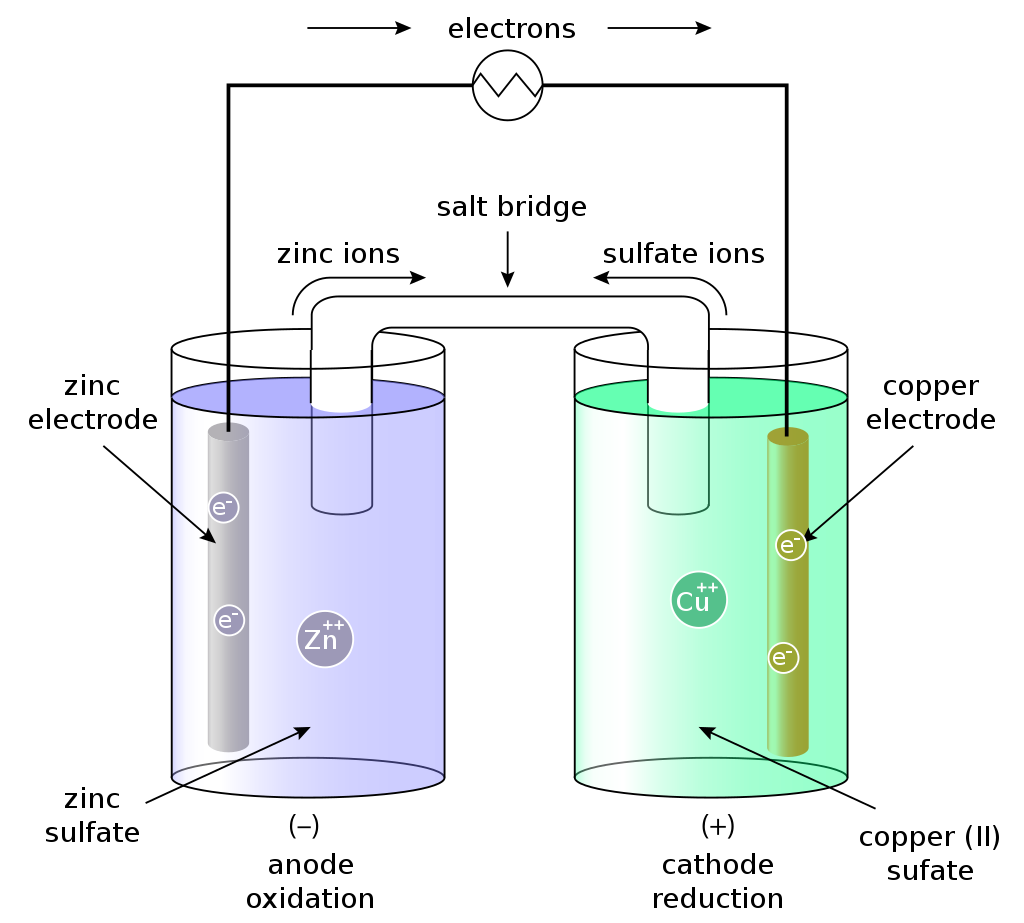
- A galvanic cell example is Daniel Cell.
- Invented in 1836 by John Frederic Danell.
- It is a type of Galvanic cell or Voltaic Cell
- The electrolyte is Copper Sulphate Solution in Copper Pot or Container dipped with a copper rod which acts as Cathode.
- Zinc rod immersed in Zinc sulphate solution which acts as an Anode.
- The two containers are connected by Salt Bridge which is a glass tube having potassium chloride or ammonium nitrate in a gelatin form.
- Salt bridge allows ionic movement but prevents mixing
- It cannot supply a steady current for a long time.
Leclanche Cell
- It produces an Emf of 1.5 V and supplies current up to 0.25 A.
- The anode is Carbon Rod.
- The Cathode is Zinc Rod.
- The electrolyte used is Ammonium Chloride.
Secondary Cells
- The anode is lead.
- The cathode is lead oxide.
- The electrolyte is Sulphuric acid.
- The advantage of a secondary cell is they are rechargeable.
- The chemical process of obtaining current from a secondary cell is called Discharge.
Electromotive Force and Internal Resistance
The emf of a battery or cell is the voltage provided by the battery when no current flows in the external circuit. The electromotive force determines the amount of work a battery or cell does to move a certain amount of charge around the circuit.
It is denoted by Symbol ξ, pronounced as Xi. An Ideal battery has zero internal resistance and the potential difference across the battery is equal to its emf.
Practically, the battery is made of electrodes and electrolytes, there will be resistance to the flow of charge within the battery. This resistance is called Internal resistance r. For a real battery, the terminal voltage is not equal to the emf of the battery.
A brand new has low internal resistance and increases with aging.
Cell in Series
Several cells can be connected to form a battery. In a series connection, the negative terminal of one cell is connected to the positive terminal of the second cell. Then the negative terminal of the third cell and so on.
The free positive terminal of the first cell and the free negative terminal of the last cell becomes the terminal of the battery. Suppose n cells, each of emf ξ volts and internal resistance r ohms is connected in series with an external resistance R.
Series connection of cells is advantageous only when the effective internal resistance of the cells is negligibly small compared with R.
Cells in Parallel
In Parallel connection, all the positive terminals of the cells are connected to one point and all the negative terminals to a second point. These two points form the positive and negative terminals of the battery.
The current to the whole battery is the same for the connection of the Parallel cells. Hence it is advantageous to connect cells in parallel when the external resistance is very small compared to the internal resistance of the cells.